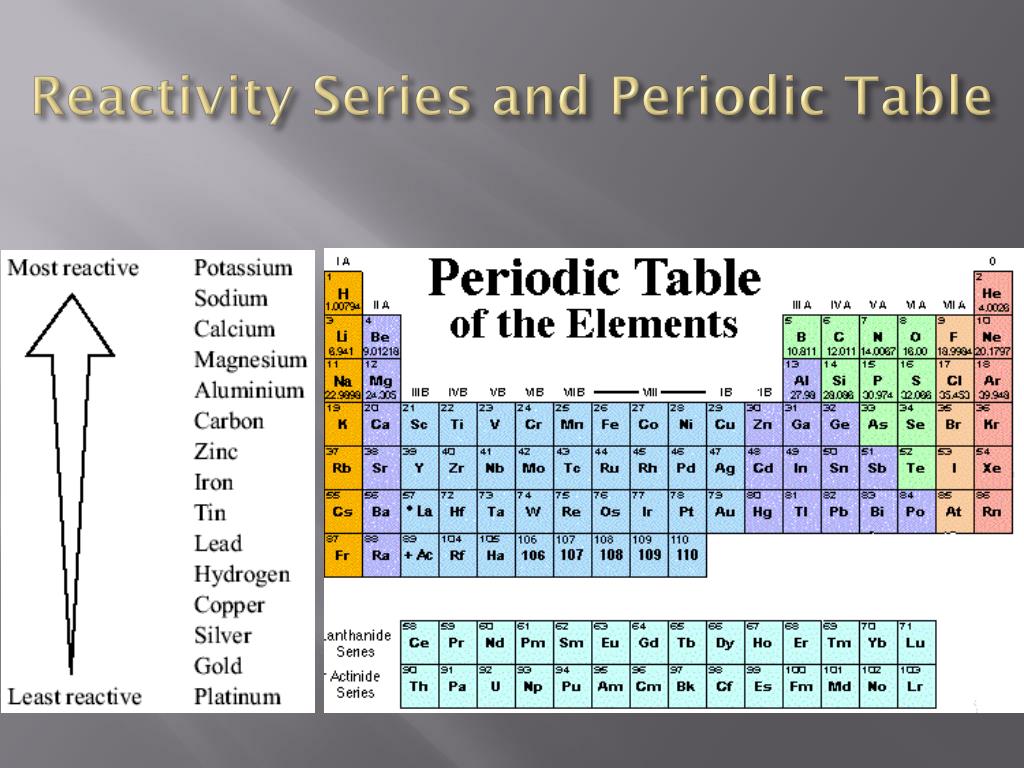 
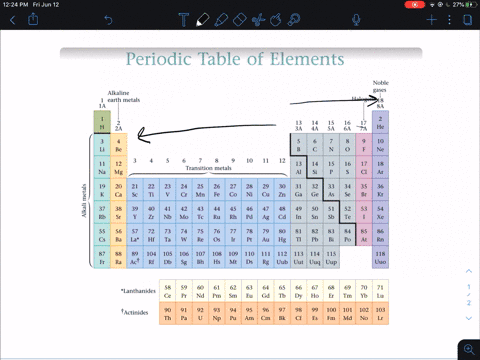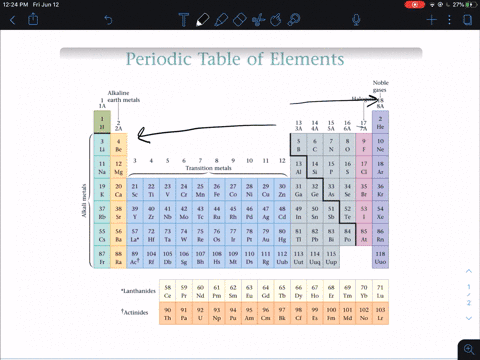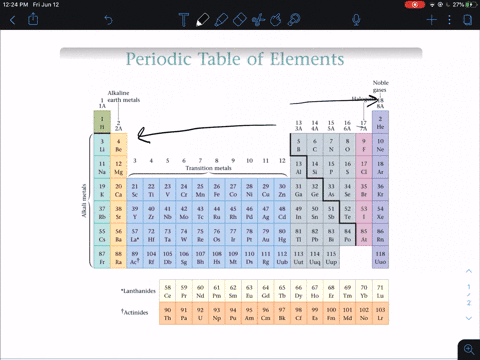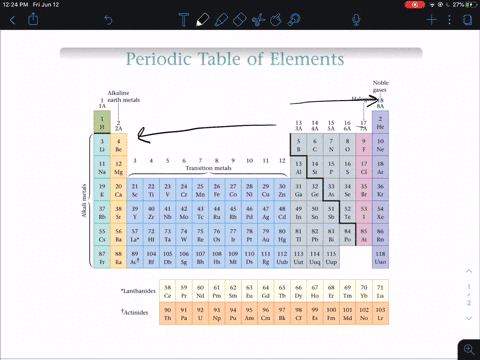
Modern Periodic Law - The properties of the elements of the modern periodic law are periodic functions of their atomic numbers. Although he predicted the properties of those elements through his periodic classification of elements. He left some space for corresponding elements in his periodic table which were not even discovered till then. Mendeleev’s Periodic Table - The arrangement of all 63 elements in rows or columns in order of their atomic weight was made by Mendeleev. Activity series of some of the more common metals, listed in descending order of reactivity. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. Newlands Law of Octaves - The elements were arranged in increasing order of their atomic weights and found that every 8 th element shows similarity with the 1 st element. The reactivity series is a series of metals, in order of reactivity from highest to lowest. In every triad, the atomic weight of the middle element was equal to the average of the atomic weights of the first and third elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
